|
Trivial names are well known for some molecular compounds and they are often used in the place of systematic names, e.g., water for H 2O and ammonia for NH 3. Metric or SI (Le Systme International d'Units) prefix are based on powers of ten.They are modifiers on the root word that tell us the unit of measure. Examples of the names are: NO is nitrogen monoxide, CO 2 is carbon dioxide, PCl 3 is phosphorous trichloride, P 2O 5 is diphosphorus pentoxide, SiO 2 is silicon dioxide. Ionic and molecular compounds are named using somewhat-different methods. The first word is the name of the first element. Table 3.5.1 lists the prefixes used to represent the number of atoms from 1 to 10. Chemists use nomenclature rules to clearly name compounds. When you have two different elements, there are usually only two words in the compound name. If the prefix ends with a vowel and the element name begins with a vowel, drop the ending vowel of the prefix.Do not write mono- if it applies to the first element in the formula, but write mono- if it applies to the second element.the name of the second element with a prefix showing the number of atoms and its last syllable replaced with –ide. First, name the nonmetal furthest to the left and bottom of the periodic table by its element name.name of the first element in the formula with a prefix showing the number of atoms, followed by,.The name of binary covalent compounds contains prefixes, listed in Table 1, to indicate the number of atoms followed by the name of the elements according to the following rules: Do not worry about the atomic number - remember, you will have a periodic table. For example, MeV means million electron volts, units of energy. Nomenclature is the naming of chemical elements and compounds. Often, the symbols are used together with units. A similar line of reasoning works with atoms on the other side of the periodic table. Prefixes - Chemistry LibreTexts Prefixes Page ID Prefixes for Decimal Multiples and Submultiples Prefixes are often used for decimal multiples and submultiples of units. For one carbon in the compound, the prefix would. 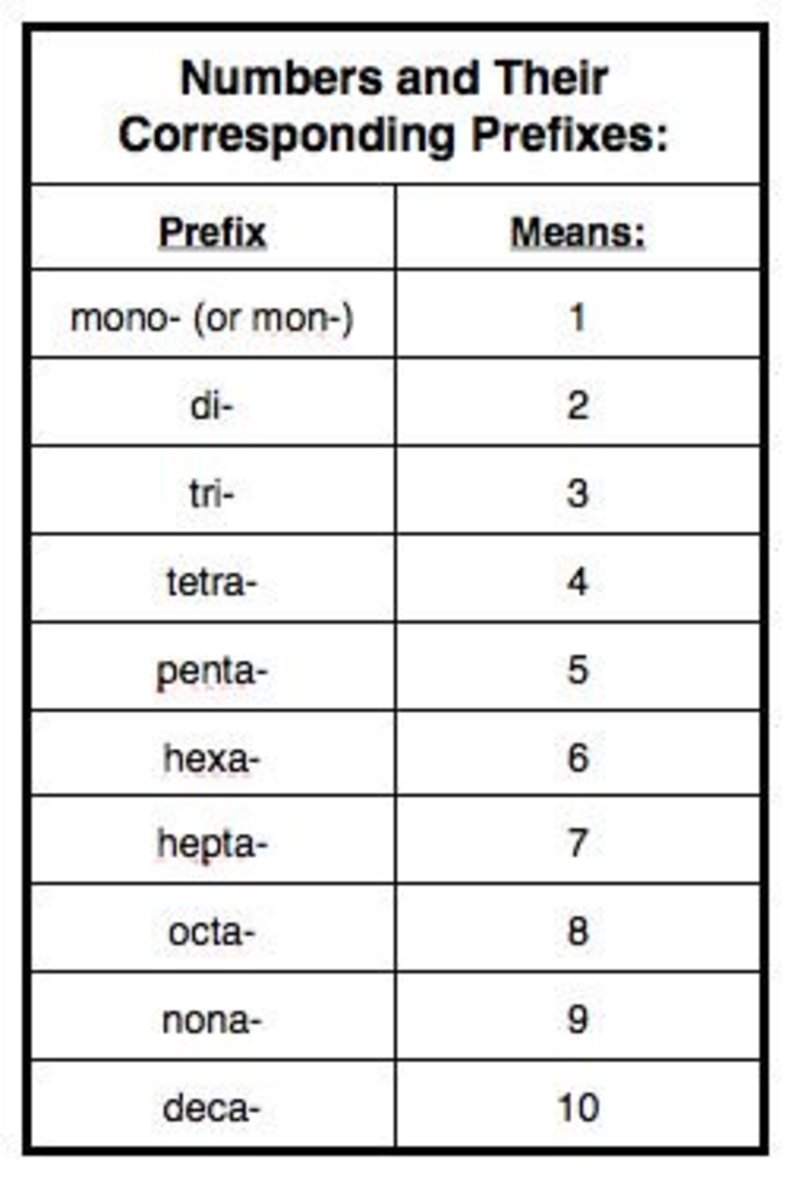
4) binary compound composed of metal ion with fixed or. Carbon is the element farther to the left in the periodic table, so carbon is the first root word in the name. e) calcium oxide f) beryllium sulfide g) sodium oxide h) lithium nitride. prefix is dropped the prefix mono- is never used for naming the first element of a compound). a) lithium fluoride b) sodium iodide c) potassium bromide d) magnesium chloride. Chemistry prefixes are used to denote the number of atoms of each element in a molecule. Numeric prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) H2AsO4 dihydrogen arsenate ion HAsO42- hydrogen arsenate ion NaH2PO4. Writing the names of binary covalent compounds Provide formulae for the following names. In order to correctly convert one metric unit to another, you will need to determine which of two prefixes represents a bigger amount and then determine the exponential "distance" between them.\) Naming covalent compounds and molecular compounds can be fun In this tutorial, you will learn how to derive the name of covalent compounds, from the molecular formula. They are modifiers on the root word that tell us the unit of measure. Metric or SI (Le Système International d'Unités) prefix are based on powers of ten. This behavior is observed for most transition metals, many actinides, and the heaviest elements of Groups 1315. Naming Binary Molecular Compounds Rule 1: If both elements in the compound are in the same period on the periodic table, the element with the smaller group. Names of hydrocarbon molecules are based on the number of carbon atoms in the molecule and the type of bond between them. As shown in Figure 2.11 'Metals That Form More Than One Cation and Their Locations in the Periodic Table', many metals can form more than one cation. Chemistry prefixes are used to denote the number of atoms of each element in a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
